A study has revealed today that yet another new coronavirus vaccine, this time by Oxford and AstraZeneca, has proven to be remarkably safe for those over the age of 60.
Discover our latest podcast
Scientists behind the vaccine have released some data from early trials which showed that the vaccine prompted strong signs of immunity in 99% of participants.
Data published in July predicting that the vaccine would only be safe for those under 55 has also been debunked. The stage two testing involving 560 people (including 240 over the age of 70) showed that the jab was effective across all age groups.
The trials so far show that people across all ages developed antibodies within 28 days of taking their first dose of the vaccine, and those antibodies were further boosted after the second dose.
However, the vaccine did also show more side effects than a placebo. Within the first week of having the vaccine, eight out of ten people under 55 claimed to suffer from arm pain, muscle aches, tiredness and headaches. Scientists claim that these side effects are considered to be mild and much more common in a younger age group.
Today, Oxford published the results of their second phase trial in the medical journal The Lancet, showing that those across all age groups showed similar immune responses.
Those taking part in the trial were split into age groups of 18-55, 56-69 and 70+ with the vaccine being better tolerated and with fewer side effects in the older age groups.
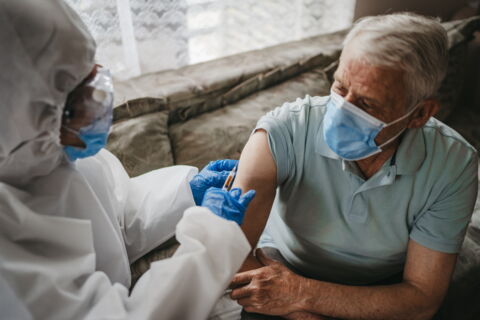
While the trials are still ongoing, the results are promising and Britain has already pre-ordered 100 million doses and if it’s effective, it could be used to protect the majority of the population.
Vaccines for those in their youth have been outlined as a lower priority than effective vaccines for the elderly as they are the ones most likely to suffer from serious health conditions and are at a higher risk of dying if infected with the virus.
Dr Gillies O'Bryan-Tear of the Faculty of Pharmaceutical Medicine explained:
The ageing of the immune system in older people can lead to lower efficacy of vaccines. This is especially important as Covid has much higher lethality in older people.
One clue to this from the current study is that older people reacted less to the vaccine – fewer fevers, less injection site pain and swelling – but encouragingly, the immune responses in the older patients were indistinguishable from those in younger patients. This suggests, but does not prove, that the protection against the illness will be similar in the elderly and the young.
This is the third vaccine to make the headlines as the Moderna vaccine and the Pfizer/BioNtech vaccine boasting a near 95% efficacy rating. However, as the Oxford vaccine is still undergoing its trial phases their efficacy results aren’t to be expected until the next few weeks.
Britain has secured doses of all three of the vaccines and up to 5 million people could be vaccinated with the Pfizer jab by the end of the year. The government has also secured 5 million doses of the Moderna vaccine which will be distributed in the early Spring.
All three vaccines will have to complete the final stages of their trials before being released to the public but so far the results seem promising and there is hope that we will soon be able to have the spread COVID-19 well under control.